
Further investigations in the 19 th century on the caffeine/HI system followed with a view to analytical aspects. In the course of the aforementioned study caffeinium triiodide monohydrate (caffHI 3 ⋅ H 2O) was identified. A reinvestigation by combustion analysis in 1865 verified these findings. CommentĪ report on the first triiodide (strychninium triiodide) dates back to the beginning of the 19 th century. All other hydrogen atoms were included using the standard riding models of the SHELX System (AFIX 43 137). 
Coordinates and U iso of the H4 hydrogen atom ( cf. The largest difference electron peaks (down to 0.51 e Å −3) are less than 0.84 Å away from iodine positions. The largest difference electron density peaks and holes are 0.99 and −0.88 e Å −3, respectively. During the latter stages of the refinement a slight disorder of the triiodide anion was taken into account (not shown in the figure). Structure solution with the SHELXT yielded all non-hydrogen atoms. 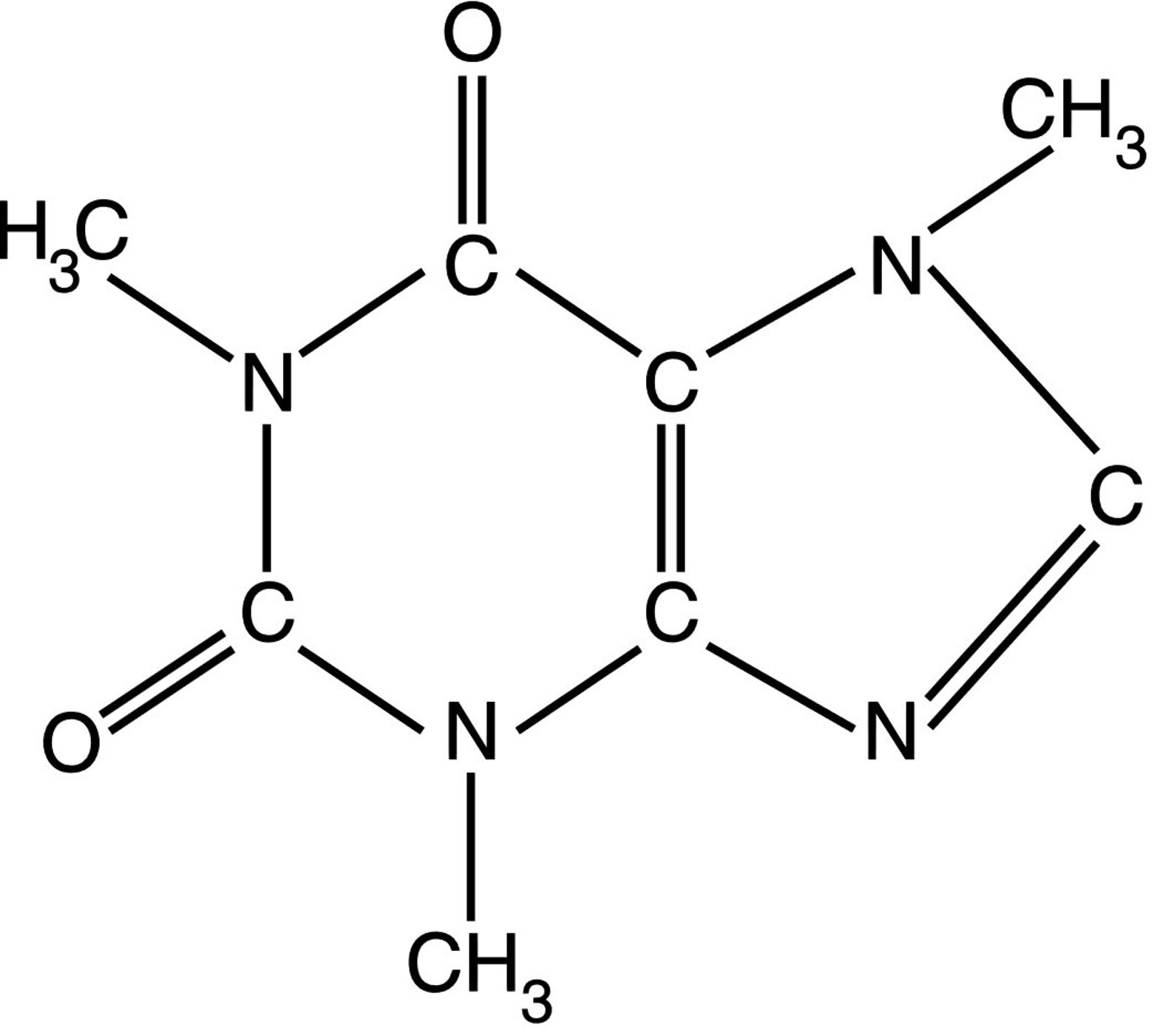
Absorption corection was applied using the SADABS program implemented in the APEX2 program system. Data collection followed the standard procedures of the Bruker APEX2 software. The crystal used for the study was harvested directly from the mother liquor. The crystals show an orange colour and are easily distinguishable from the dark caffeinium triiodide hydrate crystals. The title compound caffeinium triiodide – caffeine (1/1) was obtained from a mixing solution of ethanol and equimolar amounts of caffeinium triiodide hydrate and caffeine. Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å 2).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
